- For US Healthcare Professionals Only
- Important Safety Information
- Full Prescribing Information
- INVEGA HAFYERA™ Consumer Site
What to Do if Your Patient Misses a Dose of INVEGA HAFYERA™1
- To avoid a missed dose, patients may be given the injection up to 2 weeks before or 3 weeks after the scheduled 6-month dose
If more than 6 months and 3 weeks up to but less than 8 months have elapsed since the last injection of INVEGA HAFYERA™, do NOT administer the next dose. Instead, use this reinitiation regimen:
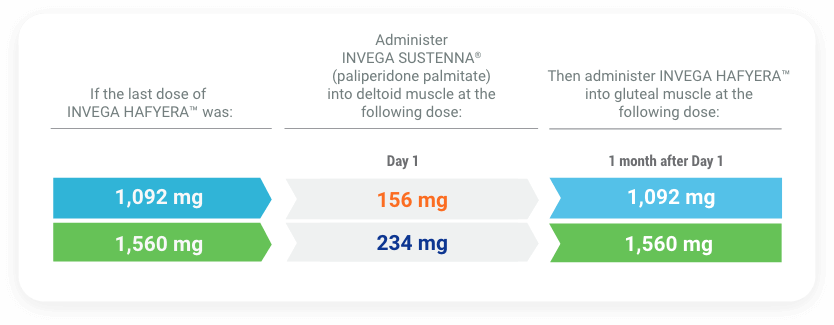
If 8 months up to and including 11 months have elapsed since the last injection of INVEGA HAFYERA™, do NOT administer the next dose. Instead, use this reinitiation regimen:

If more than 11 months have elapsed since the last injection of INVEGA HAFYERA™, reinitiate treatment with INVEGA SUSTENNA® (1-month paliperidone extended-release injectable suspension) as described in the prescribing information for that product. INVEGA HAFYERA™ can then be resumed after the patient has been adequately treated with INVEGA SUSTENNA® for at least 4 months.
REFERENCE: 1. INVEGA HAFYERA™ [Prescribing Information]. Titusville, NJ: Janssen Pharmaceuticals, Inc.; August 2021.